UDK: 616.833.34-089.5-053.2
Mikjunovikj Derebanova Lj.
1University Clinic for Traumatology, Orthopedics, Anesthesia, Reanimation, Intensive Care and
Emergency Department – Skopje; Department for Anesthesia, Reanimation and Intensive
Care – Skopje; University Clinical Center “Mother Theresa” – Skopje
Abstract
Postoperative pain management in children is quite challenging. There are numerous benefits from regional anesthesia as a part of multimodal anesthesia protocols, while failure to achieve postoperative pain control in children can cause delay in physical healing and psychological effects in the long term, such as sleep disorders, anxiety, and chronic pain. Anatomical, physiological, and pharmacological differences between adults and the pediatric population should be understood before performing regional anesthesia in children.
In the past two decades, relying on ultrasound guidance, the regional anesthesia in pediatric patients of any age has seen significant development and evolved to the point of setting a new standard. This review is based on studies, guidelines, and peer-reviewed articles focusing on pediatric regional anesthesia in current literature. This review includes descriptions and illustrations of the relevant sonoanatomy of brachial plexus on different levels, ultrasound probe positioning, surgical indications, and associated potential complications. The aim of this review hopefully, is to serve as a foundation for a better understanding of the block techniques of the upper extremity, with their clinical indications, as well as to facilitate improved perioperative pain management in children.
Key words: pediatric regional anesthesia, brachial plexus blocks, ultrasound–guided.
Introduction
Postoperative pain management in pediatric patients continues to be a significant challenge (1). Insufficient pain control may be explained by “the fear” of opioid use or inadequate analgesic use (2). The use of regional anesthesia in perioperative pain management for pediatric patients can become standardized in terms of analgesic use (3). The benefits from regional anesthesia include: lower rate of opioid complications such as respiratory depression (4,5) and decreased gastrointestinal mobility (6) or immune depression; reduced postoperative pain score; lower rate of postoperative nausea and vomiting; protective effect in reducing surgical stress and early discharge after surgery (7). Failure to achieve postoperative pain control in children can also cause delay in physical healing and psychological effects in the long term, such as sleep disorders, anxiety, and chronic pain (8). Regional anesthesia in infants improves hemodynamic stability, decreases catecholamine production, the metabolic stress response to surgery, the incidence of postoperative respiratory complications and indorses a fast return of gut function and feeding (9).
In the early 1980s, Dalens had established the first pediatric regional anesthesia teaching program in France; nevertheless its role in the pediatric anesthetic practice was boosted with the use of ultrasound (10). In 1994, Kapral and colleagues, published the first report of the use of ultrasound in regional anesthesia in adults (11). A few years later, its first application in regional anesthesia in pediatric population was reported (12). In the past two decades, relying on ultrasound guidance on the regional anesthesia in pediatric patients of any age has seen significant development and evolved to the point of setting a new standard (13,14).
This review is based on studies, guidelines and peer-reviewed articles focusing on pediatric regional anesthesia in current literature. The literature search included terms such as “pediatric regional anesthesia,” “ultrasound-guided peripheral nerve blocks,” “postoperative pain,” with an emphasis on “upper extremity peripheral nerve blocks”. To create a comprehensive narrative review of ultrasound-guided regional anesthesia used in upper extremity pediatric surgeries, a targeted literature search was conducted using PubMed, Google Scholar and Embase, focused on English-language studies addressing the safety, efficacy, and technical aspects of these techniques in pediatric patients. This review includes descriptions and illustrations of the relevant sonoanatomy of the brachial plexus at different levels. The illustrations are created by the author of this review. The sonoanatomy images were obtained using a portable ultrasound unit (Simens, Acuson P500) with linear probe, frequently used in the author’s institution for pediatric ultrasound-guided regional anesthesia. The aim of this review hopefully, is to serve as a foundation for better understanding upper extremity block techniques and clinical indications, and to facilitate improved perioperative pain management in children.
Differences Between Children and Adults
Neonates and infants have anatomical, physiological, and pharmacokinetic differences from older children and adults. The key anatomical differences include: smaller superficial nerves, vessels and tendons that lie closer together in young children. Their nerves have shorter diameter with incomplete myelination whose completion may take several years. Also, the endothelium has less connective tissue. These are the reasons why the onset of blockade is faster and the duration of regional anesthesia is usually shorter in younger children than in adults. In physiological and pharmacological aspects, infants have increased systemic absorption and accumulation of local anesthetics due to their increased cardiac output and immature hepatic function with larger distribution volumes, longer half-lives, and reduced protein binding of local anesthetics than in adults. Hepatic metabolism of local anesthetics fully matures by 9 months of age. A relatively high proportion of cardiac sodium-gated channels are in an open state, with a high affinity to local anesthetics. The main proteins to which local anesthetics bind are α1-acid glycoprotein and albumin, and their concentration is reduced by the first year of age. Therefore, the main reason for local anesthetic systemic toxicity (LAST) is the high dose of the free and hence pharmacologically active fraction of any local anesthetic (15). The risk of drug accumulation rises when a continuous infusion or multiple injections are used (16). Hence, the maximum dose for every child should be calculated individually (17).
Awake Vs Asleep
It’s a fact that children and infants are afraid of needles and can develop severe anxiety of injections, so it’s almost impossible to perform a safe block in a child who is moving. Therefore, pediatric anesthesiologists prefer to perform regional anesthesia under deep anesthesia or general anesthesia. With deep anesthesia or under general anesthesia, the child becomes “cooperative” and immobile, while complete control of the vital signs is ensured. However, when the child is under general anesthesia or deep sedation, early warning signs of complications from regional anesthesia, such as the first neurological signs of LAST or technical mistakes, would not be detectible; the test dose is not reliable, and the patient can’t report paresthesia or pain during injection. In 2014, Taenzer et al. published a report from The Pediatric Regional Anesthesia Network that included 50,000 patients and demonstrated that performing blocks while awake or under light sedation was associated with a higher incidence of postoperative neurologic symptoms compared with performing blocks under general anesthesia (rate of 6.82/1000 vs 0.93/1000, respectively) (18).
There are two main registries for complications from regional anesthesia in the pediatric population, the French-Language Society of Pediatric Anesthesiologists and Reanimateurs (Association des Anesthésistes et Reanimateurs Pédiatriques des d’Expression Francophone [ADARPEF]) and the Pediatric Regional Anesthesia Network (PRAN). In 1996 and 2010, ADARPEF reported similar complication rates of 0.09% and 0.12% respectively (95% confidence interval [CI]: 0.090.17) (19,20). Data from the PRAN registry, comprising more than 100,000 nerve blocks, confirmed similar findings (21). Transient neurological deficit was recorded in only 25 cases (2.4 in 10,000 [95% CI: 1.6-3.6 in 10,000]), but none led to permanent sequelae. The most common adverse events were catheter malfunctions (displacement, occlusion) that occurred in 4% of cases.
In 2017, the European Society of Regional Anesthesia and Pain Therapy (ESRA) and the American Society of Regional Anesthesia and Pain Medicine (ASRA) joint committee published evidence-based recommendation in which they advise performing pediatric regional anesthesia under general anesthesia or deep sedation (category B2) (22). Injection of local anesthetic should be very slow and fractionated, with frequent aspiration tests and careful ECG monitoring, particularly the T wave. When test dose of epinephrine (1mcg/kg) is used, following any ECG changes, it is recommended that in the next 90 seconds an accidental intravenous injection of local anesthetic is detected.
Ultrasound Advantages Compared to Landmark-based
Technique and Neurostimulator
Previously, high volumes of local anesthetics were used to compensate for inaccurate needle placement during landmark-based regional blocks, potentially exposing children to the risk of LAST. Ultrasonography provides additional information, including direct visualization of the neural structures to be blocked, the spread of local anesthetic around the nerve and within the appropriate fascial plane, and visualization of anatomical variations and vital structures surrounding the nerves. Sometimes, the responses to a peripheral nerve stimulator (PNS) can be unreliable and the muscle contractions elicited by these stimuli can be harmful in children with skeletal or connective tissue disorders, such as epidermolysis bullosa and syndactyly. In such cases, ultrasound guidance in preforming neural blocks prevents the need to use a PNS and reduces the risk for tissue damage (12). Studies in children and adults have shown that ultrasound-guided regional nerve blocks enable faster block performance, increase accuracy and success rates, hasten block onset, and reduce the volume of injected local anesthetics (23).
Upper Extremity Peripheral Nerve Blocks
Some of the most frequent operations in pediatric populations are fractures of the upper and lower extremities. The reported incidence of fractures is 20.2 in 1000 children per year (24). Various levels of pain have been reported in children with extremity fractures, experiencing moderate-severe pain. The most severe postfracture pain is experienced in the first 48 hours (25).
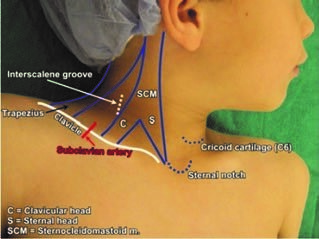
The motor and sensory innervation of the upper extremity is provided by the brachial plexus, except for the part of the shoulder, which is innervated by the cervical plexus. The medial aspect of the upper arm has sensory innervation from the intercostobrachial nerve, a branch of the 2nd intercostal nerve. The brachial plexus is formed by the anterior branches of the spinal nerve roots of C5, C6, C7, C8, and T1 and may often contain fibers from the fourth cervical and second thoracic spinal roots. Depending on the location and the type of surgery, the brachial plexus can be blocked on different levels, such as interscalene, supraclavicular, infraclavicular, and axillary blocks. The type of blocks should not be determined only by the type of surgery; it also depends on the patient characteristics and experience of the anesthesiologist who is performing the block.
Interscalene brachial plexus block (IBPB)
Indications. An interscalene block is performed for shoulder surgery or surgery of the proximal part of the humerus. Shoulder surgery is very rare in children, thus this approach to the brachial plexus is infrequently performed.
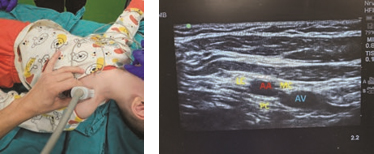
Position of the patient. The patient is placed in a supine position with the head turned contralateral to the side where the block needs to be performed (Fig. 1).
Figure 1. Position of the patient for performing the interscalene block
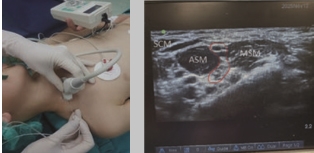
Anatomy and ultrasound characteristics. As previously mentioned, the brachial plexus is formed by the anterior branches of the spinal nerve roots of C5 – C8 and T1. The branches of these spinal nerves leave the corresponding intervertebral foramina and emerge in the interscalene groove which is formed by the anterior and middle scalene muscles. At this level, trunks are formed (the upper trunk is formed by C5 and C6, the middle trunk is a continuation of C7, and the lower trunk originates from C8 and T1). The lineal ultrasound probe is placed transversely over the neck, into the interscalene groove (proximally to the clavicula and over the external jugular vein if visible) (Fig. 2a). In small children and infants, the ultrasound probe is wide enough to capture the internal jugular vein and carotid artery together with the brachial plexus. Lateral to the great vessels lies the anterior scalene muscle and more posterolaterally lies the middle scalene muscle. Between these muscles lies the hyperechogenic interscalene sheath containing the brachial plexus (Fig 2b). The brachial plexus trunks in this section are usually visualized as more than three round or oval-shaped hypoechoic structures. In literature, this is described as sign of a “snowman” or “traffic light”.
Figure 2. a – position of the ultrasound probe and additional use of neurostimulator; b- ultrasound findings of
interscalene brachial plexus block: case of a 3-year-old child: SCM – sternocleidomastoids muscle, ASM – anterior scalene muscle, MSM – middle scalene muscle, brachial plexus is outlined with a red line
Local anesthetic and dose. 0.2% ropivacaine up to 0.5ml/kg or 0.25% bupivacaine 0.2-0.3 ml/kg (max 2mg/kg). Continuous infusion: 0.1– 0.15ml/kg/h (max 10ml/h) (26).
Complications. Possible complications in this region are intrathecal or intravascular injections of local anesthetic, phrenic nerve palsy and Horner’s syndrome.
Clinical tips.
- In infants, use hockey stick ultrasound probe.
- In-plain approach from posterior to anterior is safer then out-plane approach
- Place the needle in the interscalene sheath (between the middle and lower trunk) and follow the spreading of local anesthetic upwards, followed by frequent aspirations.
- Use color Doppler to avoid accidental vascular puncture
Supraclavicular brachial plexus block (SCBP)
Indications. The suprascapular block is performed for all upper arm surgical procedures on the upper arm and distal to the shoulder, including extensive hand, forearm, and elbow surgery. This block is often refered to as spinal anesthesia of the upper extremity. For shoulder surgery, interscalene block is considered more appropriate than the SCPB because the interscalene block also blocks the suprascapular nerve. Supraclavicular plexus block also eliminates pain caused by the tourniquet.
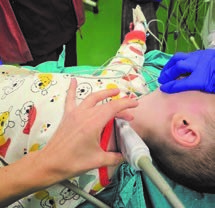
Position of the patient. The patient is placed in a supine position with the head turned contra-laterally to the side where the block needs to be performed. A pillow/roller under the shoulders should be used to achieve inline positioning of the head with the upper body (since small children have bigger heads compared to the body). The arm that needs to be blocked should be adducted to the body.
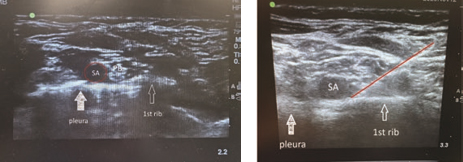
Anatomy and ultrasound characteristics. The three trunks emerging from the interscalene groove and pass downward and laterally to the subclavian artery. On this level, each trunk divides into anterior and posterior divisions. The linear probe needs to be placed coronal-oblique in the supraclavicular fossa (Fig. 3). The subclavian artery, the first rib and pleura should be visualized with the brachial plexus, which at this level appears as a hypoechogenic “cluster of grapes” posterolaterally to the subclavian artery (Fig. 4). In-plane technique is used, and the needle should be visible to avoid accidental puncture of vessels, nerves, and pleura. Always insert the needle from lateral to medial.
Figure. 3. Position of the linear probe in performing supraclavicular brachial plexus block
Figure 4. Ultrasound findings of supraclavicular brachial plexus block: pleura, first rib, SA- subclavian artery, and PB – brachial plexus posterolateral to the artery. The first image shows a US scan on a 2-year-old patient and the second an 11year old patient. The red line is directed on the “corner pocket”
Local anesthetic and dose. 0.15– 0.5 ml/kg bupivacaine 0.25% (max 2.5mg/kg) or 0.2% ropivacaine or 1% lidocaine (max 5mg/kg). In infants under 6 months of age, the dose should be halved (26).
Complications. Possible complications in this region are arterial and pleural puncture (pneumothorax), phrenic nerve palsy, and Horner’s syndrome. The latter two are more common complications in the interscalene block.
Clinical tips.
- When the patient is correctly positioned, the components of the brachial plexus become more superficial and the block’s administration in most children is easy
- Deposition of local anesthetic in “corner pocket” improves successful supraclavicular block. This region borders inferiorly by the first rib and medially by the subclavian artery (Fig. 4b) and on this level, the nerve fibers for the ulnar nerve emerge, which innervate the medial side of the arm.
- Use color Doppler to avoid accidental vascular puncture (possible presence of dorsal scapular and transverse cervical arteries that branch from the subclavian artery and pass through the brachial plexus)
Infraclavicular brachial plexus block
Indications. The infraclavicular block is performed for surgical procedures on distal humerus, elbow, forearm and hand. The infraclavicular block indications are similar to those for supraclavicular brachial plexus block.
Position of the patient. The patient is placed in a supine position with the head turned contralaterally to the side of the block needs to be performed and the arm that needs to be blocked should be positioned in adduction; the elbow should be flexed by 90° degrees and the forearm should be placed on the patient’s abdomen. A pillow/roller should be used to line the head with the upper body.
Anatomy and ultrasound characteristics. On this level, anterior and posterior divisions form lateral, medial and posterior cords, named according to their position to the axillary artery. A linear probe is placed in parasagittal plane below the clavicula and medially to the coracoid process (Fig. 5a). Beneath the pectoral muscles lies the axillary artery, which is surrounded by three cords of the brachial plexus; and the axillary vein is poisitioned medial and caudal to the artery (Fig. 5b). The cords of the plexus are visualized as hyperechoic oval structures, but sometimes the medial and posterior cords can be difficult to identify because of their anatomical variations. The in-plane technique is safer, and the needle is advanced from cranial to caudal with target point at 7-8 of the clock level under the axillary artery. At this point, deposition of the local anesthetics encircles the axillary artery in a “U-shape” and provides blocking of all three cords.
Local anesthetic and dose. 0.2– 0.3 ml/kg bupivacaine 0.25% (max 2.5mg/kg) or 1% lidocaine (max 5mg/kg). Continuous infusion – 0.4ml/kg/h (max: 8-10ml/h) (26).
Complications. Complications in this block are extremely rare, however, they may include vascular puncture, nerve injury and pneumothorax.
Figure. 5. a – position of the linear probe for performing infraclavicular brachial plexus block; b – ultrasound
findings of infraclavicular block in a 3-year-old child: AA – axillary artery, AV – axillary vein, LC – lateral cord, MC – medial cord and PC – posterior cord of the brachial plexus
Clinical tips.
- Place the linear probe medial to the coracoid process and inferior to the clavicle in parasagittal plane
- After identifying the axillary artery, advance the needle toward the posterior cord
- Sometimes, for improved visualization of the cords, abduct the arm to 90°, which brings the cords more superficially and laterally to the axillary artery.
Axillary brachial plexus block
Indications. Axillary block is performed for surgical procedures distal to the elbow.
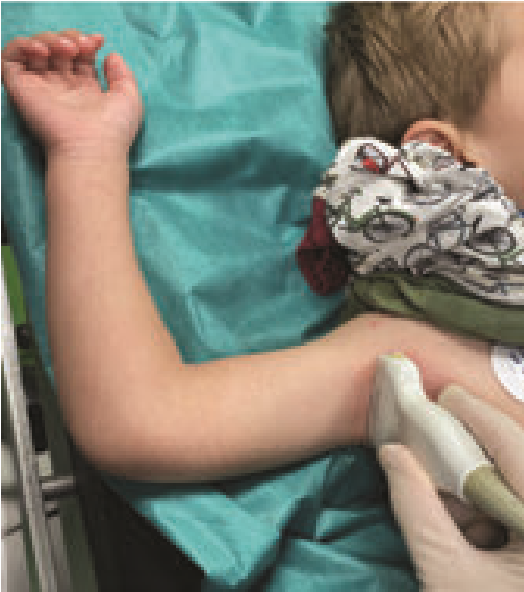
Position of the patient. The patient is placed in a supine position with the arm that needs to be blocked abducted to 90°; the shoulder should be externally rotated; the elbow should be flexed to 90° degree and the hand should be placed upwards (Fig 6).
Figure 6. Position of the patient and the linear probe for performing axillary block.
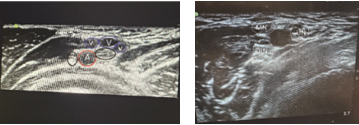
Anatomy and ultrasound characteristics. At the level of the axillary block, the median, ulnar and radial nerves are blocked. The target nerves are located in variable positions around the axillary artery, due to frequent anatomical variations (Fig. 7). The linear probe is placed in the axilla, and the target nerves are located around the anechoic axillary artery (Fig. 6 and 7). The median nerve usually lies superficially above the artery; the ulnar nerve is commonly located superficial and medial to the artery; whereas the radial nerve lies under and medial to the artery. The musculocutaneous nerve appears as a hyperechogenic oval structure between the coracobrachialis and biceps muscles. It needs to be blocked when tourniquet is applied.
Figure 7. Ultrasound findings of axillary block: A- axillary artery, V – veins, MN – medial nerve, UN – ulnar
nerve, RN – radial nerve. The first image is of a 7-year-old patient where the ulnar and medial nerves are located medially from the artery and the radial nerve is lateral to the artery. The second image is of a 15-year-old child with a different position of the nerves compared to the previous one.
Local anesthetic and dose. 0.2– 0.3 ml/kg bupivacaine 0.25% (max 2.5mg/kg) or 1% lidocaine (max 5mg/kg). 0.5 – 1ml for the musculocutaneous nerve (26).
Complications. Complications in this block are rare but may include accidental vascular puncture and, in that case, to prevent hematoma, pressure should be applied for at least 5min.
Clinical tips.
- Use color Doppler for improved visualization of the vessels in this area (usually there are more than one vein)
- For enhanced localization of the nerves, a neurostimulator may be of help.
Conclusion
Regional anesthesia as a part of the multimodal analgesia provides effective, opioid-sparing postoperative analgesia; improves comfort and hastens the recovery in children. Ultrasound imaging increases block safety, reduces complications and improves the block’s accuracy. The choice of the block should be based on surgical procedure, anatomy and experience of the anesthesiologist. In order for regional technics to be fully implemented in pediatric anesthesia, the anesthesiologist needs to undergo structured education, continuous training in the acquisition of ultrasound imaging and needling skills, and gain knowledge of pediatric anatomy and physiology as well as familiarity with the latest guidelines.
References:
- Ozen O, Saricaoglu F. Current approaches in pediatric regional anesthesia. JARSS 2025;33(2):83-93.
- Boric, K.; Dosenovic, S.; Kadic, A.J.; Batinic, M.; Cavar, M.; Urlic, M.; Markovina, N.; Puljak, L. Interventions for postoperative pain in children: An overview of systematic reviews. Pediatr. Anesth. 2017, 27, 893–904.
- Schug, S.A.; Chong, C. Pain management after ambulatory surgery. Curr. Opin. Anesthesiol. 2009, 22, 738–743
- Cullen KA, Hall MJ, Golosinskiy A. Ambulatory surgery in the United States, 2006. Bethesda (MD): National Center for Health Statistics; 2009. p. 1–25.
- Niesters M, Overdyk F, Smith T, Aarts L, Dahan A: Opioid-induced respiratory depression in paediatrics: A review of case reports. Br J Anaesth 2013; 110:175 82. doi:10.1093/bja/ aes447
- Russell P, von Ungern-Sternberg BS, Schug SA: Perioperative analgesia in pediatric surgery. Curr Opin Anaesthesiol 2013; 26:420–7. doi: 10.1097/aco.0b013e3283625cc8
- Burd RS, Cartwright JA, Klein MD: Factors associated with the resolution of postoperative ileus in newborn infants. Int J Surg Investig 2001; 2:499–502.
- Sacerdote P, Franchi S, Panerai AE: Non-analgesic effects of opioids: mechanisms and potential clinical relevance of opioid-induced immune depression. Curr Pharm Des 2012; 18:6034–42. doi:10.2174/138161212803582496
- Power, N.M.; Howard, R.F.; Wade, A.M.; Franck, L.S. Pain and behaviour changes in children following surgery. Arch. Dis. Child. 2012, 97, 879–884.
- Bosenberg A. Regional anaesthesia in children: an update. South Afr J Anaesth Analg 2013; 19: 282-8
- Dalens B. Regional anesthesia in children. Anesth Analg 1989; 68: 654-72
- Kapral S, Krafft P, Eibenberger K et al. Ultrasound-guided supraclavicular approach for regional anesthesia of the brachial plexus. Anesth Analg 1994; 78: 507-13
- Marhofer P, Sitzwohl C, Greher M et al. Ultrasound guidance for infraclavicular brachial plexus anesthesia in children. Anaesthesia 2004; 59: 642-6
- Marhofer P, Greher M, Kapral S: Ultrasound guidance in regional anaesthesia. Br J Anaesth 2005; 94:7–17. doi:10.1093/bja/aei002,
- Lam DKM, Corry GN, Tsui BCH: Evidence for the use of ultrasound imaging in pediatric regional anesthesia: A systematic review. Reg Anesth Pain Med 2016; 41:229–41. doi:10.1097/AAP.0000000000000208
- Mazoit J-X, Dalens BJ: Pharmacokinetics of local anaesthetics in infants and children. Clin Pharmacokinet 2004; 43:17–32. doi:10.2165/00003088-200443010-00002
- Muhly WT, Gurnaney HG, Ganesh A. Regional anesthesia for pediatric knee surgery: A review of the indications, procedures, outcomes, safety, and challenges. Local Reg Anesth 2015;8:85-91
- Jöhr M. Regional anaesthesia in neonates, infants and children: An educational review. Eur J Anaesthesiol 2015;32(5):289-97
- Taenzer AH, Walker BJ, Bosenberg AT, et al. Asleep versus awake: does it matter?: Pediatric regional block complications by patient state: A report from the Pediatric Regional Anesthesia Network. Reg Anesth Pain Med 2014;39(4):279 83.
- Giaufre E, Dalens B, Gombert A. Epidemiology and morbidity of regional anesthesia in children: a one-year prospective survey of the French-Language Society of Pediatric Anesthesiologists. Anesth Analg 1996; 83: 904-12
- Ecoffey C, Lacroix F, GiaufreE et al. Epidemiology and morbidity of regional anesthesia in children: a follow-up one-year prospective survey of the French-Language Society of Paediatric Anaesthesiologists (ADARPEF). Paediatr Anaesth 2010; 20: 1061-9
- Walker BJ, Long J, Madhankumar S et al. Complications in pediatric regional anesthesia: an analysis of more than 100,000 blocks from the Pediatric Regional Anaesthesia Network. Anesthesiology 2018; 129: 721-32
- Lönnqvist PA, Ecoffey C, Bosenberg A, Suresh S, Ivani G. The European society of regional anesthesia and pain therapy and the American society of regional anesthesia and pain medicine joint committee practice advisory on controversial topics in pediatric regional anesthesia I and II: what do they tell us? Curr Opin Anaesthesiol 2017;30(5):613-20
- Boretsky KR. Regional anesthesia in pediatrics: marching forward. Curr Opin Anaesthesiol 2014; 27: 556-60, Marhofer P, Harrop-Griffiths W, Kettner SC et al. Fifteen years of ultrasound guidance in regional anaesthesia: part 1. Br J Anaesth 2010; 104: 538-46
- Rennie, L.; Court-Brown, C.M.; Mok, J.Y.; Beattie, T.F. The epidemiology of fractures in children. Injury 2007, 38, 913–922.
- Drendel, A.L.; Lyon, R.; Bergholte, J.; Kim, M.K. Outpatient pediatric pain management practices for fractures. Pediatr. Emerg. Care 2006, 22, 94–99.