UDK: 616-089.843:616-036.88-052
Gavrilovska Brzanov A.
1University Clinic for Traumatology, Orthopedic Disease, Anesthesiology, Reanimation and Intensive Care Medicine and Emergency Department, Faculty of Medicine, Ss. Cyril and
Methodius University, Skopje, R.N Macedonia
Abstract
Optimal management for a potential deceased donor is crucial to the transplantation process, as it significantly influences both the quantity and quality of organs available for transplantation. Upon determination of death by neurological criteria (DNC), the body undergoes significant physiological alterations, including hemodynamic instability, endocrine failure, poor thermoregulation, hypernatremia, metabolic acidosis, and coagulopathy. The primary objective of donor management is to restore homeostasis by ensuring sufficient blood flow and oxygen supply to the organs, thereby mitigating ischemic harm and enhancing the likelihood of transplant survival.
Hemodynamic stability remains a central determinant of organ viability in the potential deceased donor. Targeting key hemodynamic and vital parameters through goal-directed therapy is essential for maintaining adequate organ perfusion. Continuous or intermittent monitoring—using available techniques such as invasive arterial pressure, central venous pressure, or advanced cardiac output monitoring—guides individualized therapy. Fluid resuscitation should primarily rely on balanced crystalloids, among which Ringer’s or Plasma-Lyte solutions are preferred over normal saline due to their favorable acid–base profile and reduced risk of hyperchloremic acidosis. The use of colloids is generally discouraged; however, in cases unresponsive to crystalloids, albumin may be considered. When adequate perfusion cannot be achieved with fluids alone, vasopressors and inotropes should be titrated based on clinical response. Norepinephrine is preferred for maintaining mean arterial pressure due to its predictable α-adrenergic effect, while dobutamine is the inotrope of choice in donors with myocardial dysfunction, enhancing contractility and cardiac output. Vasopressin may be added as an adjunct agent because of its stable vasomotor action, absence of cardiotoxicity, and catecholamine-sparing effect, particularly in donors with diabetes insipidus. Low-dose dopamine (≤3μg/kg/min) may be considered selectively in hemodynamically stable donors, as some studies suggest a potential renal-protective and immunomodulatory benefit; however, its use is controversial and not routinely recommended due to the risk of arrhythmias and metabolic instability.
Endocrine replacement therapy, including methylprednisolone (15 mg/kg), triiodothyronine (T3), and desmopressin (DDAVP), is incorporated in several donor management protocols to enhance hemodynamic stability, preserve hormonal balance, and attenuate the inflammatory cascade. Desmopressin is preferred for the treatment of diabetes insipidus due to its selective V₂-receptor activity, effective antidiuretic action, and lack of vasoconstrictive or cardiotoxic effects. While the routine use of thyroid hormone supplementation remains debated, corticosteroids play a crucial role in restoring vascular responsiveness, improving cardiac output, and reducing cytokine-mediated inflammation following brain death.
Protective mechanical ventilation (tidal volume 6–8 mL/kg, PEEP 8–10 cmH₂O, PaO₂/FiO₂ >300) should be maintained to prevent barotrauma and volutrauma. Early administration of corticosteroids and bronchodilators (salbutamol or terbutaline) helps improve alveolar fluid clearance and lung preservation. Prevention of hypothermia through active warming (maintaining core temperature above 35°C) reduces arrhythmias, coagulopathy, and metabolic acidosis. Nutritional support, parenteral or controlled enteral feeding, should continue up to the time of organ retrieval to maintain metabolic balance and prevent catabolism.
Continuous, multidisciplinary coordination among intensivists, anesthesiologists, and transplant coordinators is vital for donor stabilization and optimal organ perfusion. Any delay or inconsistency in donor management increases the risk of secondary ischemic injury and reduces the number of viable grafts. In modern transplant medicine, maintaining perfusion in the deceased donor is not merely the continuation of intensive care but a deliberate strategy to preserve organ function. Implementation of standardized donor management algorithms, close monitoring of oxygenation and hemodynamic parameters, and targeted hormonal stabilization have proven essential for achieving successful multi-organ procurement and improving post-transplant outcomes
Keywords: potential deceased donor; donor by neurological criteria; organ perfusion; donor management.
Introduction
Organ transplantation constitutes a definitive life-sustaining treatment for patients with endstage organ failure. Nevertheless, the disparity between organ demand and supply continues to widen worldwide. Organ donation primarily occurs after the determination of death by neurological criteria (DNC) or, in an increasing number of countries, after the determination of death by circulatory criteria (DCD). While both pathways are recognized within international frameworks, their implementation varies according to national legislation, clinical infrastructure, and organizational maturity of the transplant program (1-3).
In the Republic of North Macedonia, organ donation and transplantation are currently performed through living donation and deceased donation following DNC. Donation after circulatory death has not yet been introduced into clinical practice, although it is recognized as a strategic future step for expanding the donor pool and aligning with European trends (4).
Brain death induces profound systemic disturbances, including cardiovascular instability, endocrine dysregulation, inflammatory activation, and loss of thermoregulatory control. Without targeted physiological support, these disruptions jeopardize organ viability, reducing both the number and quality of transplantable grafts. Therefore, optimal donor management is understood as a continuation of intensive therapy, shifting the clinical objective from neurological recovery to the preservation of organ perfusion, oxygen delivery, and metabolic homeostasis (5).
International guidelines, including the Council of Europe EDQM Guide to the Quality and Safety of Organs for Transplantation (9th Edition), emphasize that structured, protocol-driven donor management measurably increases organ utilization rates and improves graft survival outcomes. These principles form the foundation of donor care within our national practice as well (1,6).
Pathophysiological Changes Following Brain Death
The transition to death determined by neurological criteria (DNC) precipitates a complex cascade of pathophysiological alterations that affect multiple organ systems simultaneously. The initial phase is characterized by a profound autonomic disinhibition, wherein abrupt and massive sympathetic discharge leads to marked hypertension, tachyarrhythmias, and increased myocardial oxygen consumption. This catecholaminergic surge may induce myocardial stunning, mediated by β-adrenergic receptor downregulation and subendocardial ischemia, leading to reduction in left ventricular systolic function (1,7).
Subsequently, as brainstem integrity deteriorates, sympathetic tone collapses, resulting in systemic vasodilation, vasoplegic shock, decreased coronary perfusion pressure, and declining cardiac output. The combination of reduced preload, impaired vascular responsiveness, and myocardial dysfunction significantly compromises global organ perfusion (1,8).
Simultaneously, disruption of the hypothalamic–pituitary axis results in marked endocrine dysfunction. The most frequent consequence is diabetes insipidus (DI), manifesting as excessive polyuria, hypovolemia, and hypernatremia, which directly threatens renal, hepatic, and cardiac graft integrity. Additionally, adrenal insufficiency leads to inadequate cortisol-mediated vascular support, further blunting the physiological stress response. Reduced circulating concentrations of triiodothyronine (T3) and thyroxine (T4) impair cellular oxygen utilization, myocardial contractility, and thermogenic capacity, thereby worsening hemodynamic instability (1).
In parallel, DNC triggers a systemic inflammatory response syndrome (SIRS). Pro-inflammatory cytokines increase endothelial permeability, contributing to interstitial edema, particularly within the lungs, where it may progress to neurogenic pulmonary edema and reduced oxygenation capacity. At the microcirculatory level, dysregulated nitric oxide and catecholamine signaling contribute to capillary maldistribution and impaired tissue oxygen delivery, further jeopardizing graft viability (1,2).
Loss of central thermoregulatory control leads to progressive hypothermia, which exacerbates coagulopathy, arrhythmogenic vulnerability, and reduced metabolic efficiency. When uncorrected, hypothermia accelerates the decline of organ function in the hours following DNC (1).Collectively, these alterations illustrate that brain death is not a hemodynamically stable state, but rather a dynamic and rapidly evolving pathophysiological syndrome. Prompt, targeted, and protocol-driven stabilization is therefore essential to preserve adequate organ perfusion, oxygenation, and metabolic homeostasis, ensuring optimal graft quality and transplantation outcomes (1).
Table 1. Incidence of Pathological alteration
| Pathological alteration | Incidence (%) |
| Hypotension | 81–97 |
| Diabetes insipidus | 46–78 |
| Disseminated intravascular coagulation (DIC) | 29–55 |
| Pathological alteration | Incidence (%) |
| Cardiac arrhythmias | 25–32 |
| Pulmonary edema | 13–18 |
Management Strategy
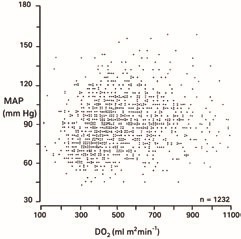
Hemodynamic monitoring is essential for donor stabilization; nevertheless, no single parameter offers a comprehensive depiction of circulatory sufficiency. Conventional monitoring techniques in critical care and perioperative medicine—such as non-invasive blood pressure, heart rate, and urine output measurements—are prevalent due to their accessibility. However, each variable is affected by physiological and non-physiological factors that undermine their reliability as standalone indicators of effective organ perfusion. Arterial pressure may indicate variations in systemic vascular resistance more than actual intravascular volume or cardiac output, whereas urine output is affected by neurohormonal regulation, renal concentrating capacity, and pharmacological agents rather than just renal perfusion (1,3). Crucially, sustaining a “normal” mean arterial pressure (MAP) does not inherently correspond to sufficient oxygen delivery (DO₂) to tissues. Research involving sedated patients and individuals having significant vascular surgeries reveals an inconsistent linear correlation between mean arterial pressure (MAP) and oxygen delivery (DO₂), mostly attributable to fluctuating microvascular tone and varying organ autoregulatory thresholds. In this scenario, tissue hypoperfusion may persist despite an appropriate mean arterial pressure, a phenomenon known as hidden or occult hypoperfusion. Clinically, this indicates that the assessment of perfusion adequacy should rely on dynamic or integrative signs rather than on pressure readings solely, including lactate trends, venous oxygen saturation, and peripheral perfusion indices (1,9).
Figure 1. Correlation of O₂ delivery (DO₂) with mean arterial pressure (MAP) in the perioperative period in patients undergoing abdominal aortic surgery (9).
The aim of hemodynamic monitoring in potential deceased donors is to sustain adequate systemic blood flow and oxygen delivery to maintain cellular metabolism, rather than to normalize isolated pressure metrics. Modern monitoring techniques can be classified based on their level of invasiveness and the nature of the information they yield. Invasive techniques, such the pulmonary artery catheter and transpulmonary thermodilution systems, provide direct evaluation of cardiac output and volumetric preload indicators. Minimally invasive systems, such as pulsewave contour analysis or lithium dilution, provide continuous cardiac output monitoring with reduced procedural complexity. Non-invasive techniques, like targeted cardiac ultrasonography and thoracic bioimpedance, provide significant real-time information on ventricular filling and contractility without the need for vascular hardware (1).
The selection of monitoring modality is contingent upon institutional resources, physician proficiency, and donor stability. Irrespective of the technique employed, therapy must adhere to a goal-oriented methodology, focusing on perfusion optimization rather than attainment of arbitrary numerical benchmarks.
Table 2. Target Parameters for Optimizing Organ Perfusion
| Parameter | Clinical Objective | Interpretation Note |
| MAP 65–75 mmHg | Supports coronary and renal perfusion | MAP alone does not reflect DO₂; always evaluate trends and context |
| CVP 6–10 mmHg | Helps guide volume status | Extreme values are more informative than single measurements |
| Lactate (trend) | Marker of global perfusion | A downward trend is more meaningful than a single value |
| Urine output 0.5–3 mL/kg/h | Surrogate of renal perfusion if DI is excluded | Interpret together with sodium and serum osmolality |
Fluid Therapy
Fluid therapy in a possible deceased donor should focus on restoring and maintaining sufficient intravascular volume to support cardiac output and organ perfusion, while preventing fluid excess and metabolic disturbances. Balanced crystalloids, including Ringer’s Lactate or Plasma-Lyte, are preferred as primary solutions due to their more physiological electrolyte composition and reduced risk of inducing hyperchloremic metabolic acidosis, a complication frequently linked to the extensive infusion of 0.9% sodium chloride. This acidity may hinder renal blood flow and microcirculatory function, making it undesirable in the donor context (1,9).
When crystalloids alone fail to attain hemodynamic stability, albumin solutions may be judiciously administered to enhance intravascular oncotic pressure and facilitate volume expansion. Albumin 4% is used for intravascular volume replenishment in cases of suspected relative hypovolemia or fluid redistribution into the interstitial compartment. Conversely, albumin 20% may be indicated in cases of pronounced hypoalbuminemia or systemic edema, with the therapeutic objective of redistributing interstitial fluid into the vascular compartment and alleviating tissue congestion, particularly in the pulmonary system. The administration must be meticulously titrated to prevent a rapid increase in intravascular volume that may aggravate cardiac dysfunction (9). Synthetic colloids, such as hydroxyethyl starch solutions, are typically eschewed because of their recognized correlation with renal impairment, tissue accumulation, coagulopathy, and inferior post-transplant graft outcomes. Their application offers no benefit compared to crystalloid treatment in this clinical situation and may be detrimental to transplantable organs (1). The entire strategy advocates for a methodical, objective-oriented therapy – utilizing balanced crystalloids as the cornerstone of volume resuscitation, thereafter employing albumin selectively when crystalloids fail to ensure stable perfusion and sufficient organ oxygenation (9).
Vasoactive Support
When optimization of intravascular volume fails to maintain adequate perfusion and organ oxygenation, vasoactive drugs are administered in a systematic, objective-driven approach. The choice of agent is contingent upon the primary hemodynamic disturbance, including vasoplegia, diminished cardiac contractility, or catecholamine resistance (1). Norepinephrine is typically advised as the primary vasopressor for donors experiencing vasodilatory hypotension. It commences at 0.02–0.2 μg/kg/min and is adjusted based on the desired mean arterial pressure and overall perfusion condition. Its primarily α-adrenergic impact elevates systemic vascular resistance and enhances coronary perfusion without notable chronotropic stimulation (1,10).
Dobutamine is the preferred inotropic drug for cardiac dysfunction, particularly after myocardial shock linked to the initial catecholamine spike in brain death. It is often administered at a dosage of 2–10 μg/kg/min, to enhance stroke volume and cardiac output. Meticulous titration is essential to prevent tachyarrhythmia and elevated myocardial oxygen demand.
Vasopressin is crucial for donors exhibiting catecholamine-resistant vasoplegia or concurrent DI. In these cases, vasopressin enhances vascular tone via V1 receptor-mediated vasoconstriction while concurrently diminishing polyuria through V2 receptor activation in the kidney. It is typically supplied at a rate of 0.01–0.04 units per minute via continuous infusion. Crucially, at these dosages, vasopressin does not induce direct cardiotoxicity nor elevate myocardial oxygen demand, rendering it particularly advantageous for donors with cardiac susceptibility (1,11).’
Low-dose dopamine, once utilized to enhance renal perfusion, is now typically eschewed due to its arrhythmogenic risks, unknown endocrine consequences, and little evidence for improving transplant outcomes. Consequently, its routine application in donor management is not advised. The primary objective is to attain hemodynamic stability while reducing myocardial stress and maintaining microcirculatory flow, with vasoactive therapy meticulously included with fluid and endocrine management techniques (1,11).
Endocrine Stabilization
Endocrine disorders frequently occur after DNC, primarily because of hypothalamic-pituitary dysfunction. DI is treated with desmopressin (DDAVP 1–4 μg IV) and meticulous free water replenishment to prevent hypernatremia and hypovolemia. If hypotension continues despite sufficient fluids and vasopressors, adrenal insufficiency must be evaluated, and methylprednisolone 15 mg/kg IV should be given to enhance vascular reactivity. While diminished T3/T4 levels are common, routine thyroid hormone replacement is not advised and should be reserved solely for refractory cardiac failure. Corticosteroid medication also provides anti-inflammatory benefits that contribute to the maintenance of lung function for transplantation (11).
Temperature Management
Hypothermia (core temperature < 35°C) commonly results from impairment of central thermoregulation following DNC. The fall in body temperature correlates with diminished metabolic activity and oxygen utilization, while leading to reduced cardiac output, heightened risk of arrhythmias, and systemic hypoperfusion. The stiffness of erythrocyte membranes increases at lower temperatures, obstructing microcirculatory flow and reducing effective oxygen delivery at the tissue level. Moreover, hypothermia induces coagulopathy by impeding enzymatic processes in the coagulation cascade.
Consequently, the preservation of normothermia (≥ 36°C) is advised in standard donor management, generally accomplished through active warming techniques including heated airway circuits, fluid warmers, and external warming blankets. Evidence indicates that modest therapeutic hypothermia may provide distinct advantages in certain donors, particularly by mitigating ischemia-induced renal damage and enhancing early graft function in kidney transplantation. Continuous clinical assessment is enhancing the equilibrium between normothermic stabilization and regulated mild hypothermia in organ-specific preservation techniques (1,12).
Lung-Protective Ventilation
Mechanical ventilation in the potential deceased donor should follow a lung-protective strategy in order to preserve alveolar integrity and optimize oxygenation while preventing ventilator-induced lung injury. Tidal volumes are generally maintained between 6 and 8 mL/kg of predicted body weight, with the application of moderate positive end-expiratory pressure (PEEP 8–10 cmH₂O) to support alveolar recruitment and prevent atelectasis. Peak airway pressures are kept below 35 mmHg to limit barotrauma. A closed suctioning system is preferred to reduce derecruitment, and recruitment maneuvers may be used intermittently when oxygenation declines. Inspired oxygen concentration is titrated to the lowest FiO₂ capable of maintaining adequate oxygenation, aiming for PaO₂ of 80–100 mmHg and a PaO₂/FiO₂ ratio greater than 300 mmHg, while maintaining PaCO₂ between 35 and 45 mmHg and arterial pH of 7.3–7.35 (1,13,14).
Supportive measures further contribute to improved lung preservation. These include adequate humidification of inspired gases, the use of mucolytics when secretions are viscous, and bronchoscopy for airway clearance when needed. Nursing strategies such as elevating the head of the bed to 30°, periodic repositioning every two hours, diligent oral care, and repeated imaging to monitor pulmonary status help to reduce ventilator-associated complications and maintain lung suitability for transplantation (1,13).
Nutritional and Metabolic Support
Globally, there is no standardized policy regarding donor feeding or fasting, with practices differing according to institutional standards and donor clinical stability. Enteral nutrition should be sustained whenever possible, to ensure metabolic substrate availability and support cellular homeostasis. Preventing prolonged hunger is crucial, as heightened catabolism may jeopardize graft healing and post-transplant functionality (15,16).
Hemostasis and Coagulation Management
Disruption of the cerebral tissue during the progression to death by neurological criteria can trigger systemic coagulation abnormalities, ranging from disseminated intravascular coagulation (DIC) to hyperfibrinolysis. These alterations are further intensified by hypoperfusion, acidosis, and inflammatory activation. Early recognition and correction are essential to preserve organ perfusion and reduce the risk of intraoperative bleeding during retrieval.
Key laboratory targets generally include: Platelet count > 50 × 10⁹/L; Fibrinogen > 1 g/L; INR < 1.5. Hemoglobin and hematocrit thresholds should be individualized based on donor age, comorbidity profile, and evidence of impaired oxygen delivery (e.g., reduced SvO₂, rising lactate). When transfusion is required, leukocyte-depleted blood products are recommended to reduce the risk of cytomegalovirus transmission and inflammatory graft injury (1,14).
Thromboprophylaxis
Thromboembolic risk remains clinically relevant, particularly in donors maintained in intensive care for prolonged periods. Thromboprophylaxis should include: mechanical measures (e.g., intermittent pneumatic compression) in all donors and/or pharmacological prophylaxis (low molecular weight heparin or unfractionated heparin) unless contraindicated because of active bleeding risk or uncontrolled coagulopathy. This balanced approach aims to maintain effective microcirculatory flow while minimizing hemorrhagic complications during organ procurement (1).
Antimicrobial Treatment
Microbiological monitoring and prompt anti-infective intervention are essential elements of standard donor management. Upon identifying a suitable donor, blood, urine, tracheal aspirate, and, where appropriate cerebrospinal fluid, cultures should be collected, with additional sampling directed by clinical progression. Upon confirmation or high suspicion of infection, antimicrobial therapy should be promptly commenced and tailored based on microbiological results, prioritizing drugs that ensure sufficient tissue penetration.
In donors lacking clinical, laboratory, or imaging indicators of infection and who are not undergoing antimicrobial therapy, routine antibiotic prophylaxis is not advised, as it has not shown efficacy in enhancing graft outcomes and may foster antimicrobial resistance (1,17,18).
Organ Procurement
The concluding stage of donor management involves the organization and implementation of organ procurement. The donor’s physiological state during retrieval directly influences graft survivability and post-transplant functionality. The key goal during this time is to maintain sufficient organ perfusion, oxygen transport, and metabolic equilibrium.
In hemodynamically stable donors, multiple studies suggest that a controlled delay in organ procurement (exceeding 20 hours post-confirmation of stabilization) may facilitate partial recovery of organ function by enhancing microcirculatory regulation and mitigating the inflammatory response. Such a technique should only be contemplated when donor stability is maintained and logistical conditions permit synchronized retrieval without the risk of secondary deterioration.
Upon finalization of consent and planning of the retrieval procedure, delays must be minimized to prevent growing hemodynamic instability. It is crucial to sustain normovolemia, adequate oxygenation, and sufficient cardiac output throughout the procurement process until aortic cross-clamping occurs. Sudden hemodynamic alterations, hypoxia, or hypovolemia during the immediate pre-retrieval phase can jeopardize transplant quality (1).
Conclusion
Donor management following DNC is a proactive, protocol-driven extension of intensive care, focused on maintaining organ perfusion and cellular equilibrium. Structured hemodynamic optimization, endocrine stabilization, lung-protective ventilation, temperature regulation, metabolic support, and coordinated interdisciplinary care are crucial for maximizing organ utilization and enhancing post-transplant outcomes.
References:
- European Directorate for the Quality of Medicines & HealthCare (EDQM). Guide to the quality and safety of organs for transplantation. 9th ed. Strasbourg: Council of Europe; 2025.
- DuBose J, Salim A. Aggressive organ donor management protocol. J Intensive Care Med 2008; 23:367-75.
- Wood KE, Becker BN, McCartney JG, et al. Care of the potential donor. N Engl J Med. 2004; 351:2730–2739.
- Kuzmanovska B, Gavrilovska Brzanov A. Challenges in the program of deceased donor transplantation in North Macedonia. UTSAK. 2024;17:285–289..
- Conrick-Martin I, Gaffney A, Dwyer R, et al. Intensive Care Society of Ireland guidelines for management of the potential organ donor. Ir J Med Sci. 2019;188:1111–1118..
- Kutsogiannis DJ, Pagliarello G, Doig C et al. Medical management to optimize donor organ potential: review of the literature. Can J Anaesth 2006; 53:820-30.
- Zaroff JG, Babcock WD, Shiboski SC et al. Temporal changes in left ventricular systolic function in heart donors: results of serial echocardiography. J Heart Lung Transplant 2003; 22:383-8.
- Martin-Loeches I, Sandiumenge A, Charpentier J et al. Management of donation after brain death (DBD) in the ICU: the potential donor is identified, what’s next? Intensive Care Med 2019; 45:322-30.
- Reinhart K, Perner A, Sprung CL, et al. European Society of Intensive Care Medicine. Consensus statement of the ESICM task force on colloid volume therapy in critically ill patients. Intensive Care Med. 2012 Mar;38(3):368-83. doi: 10.1007/s00134-012-2472-9.
- Benck U, Gottmann U, Hoeger S et al. Donor desmopressin is associated with superior graft survival after kidney transplantation. Transplantation 2011; 2011; 92:1252-8.
- Dupuis S, Amiel JA, Desgroseilliers M et al. Corticosteroids in the management of braindead potential organ donors: a systematic review. Br J Anaesth 2014; 113:346-59.
- Malinoski D, Saunders C, Swain S et al. Hypothermia or machine perfusion in kidney donors. N Engl J Med 2023; 388:418-26.
- Arjuna A, Mazzeo AT, Tonetti T et al. Management of the potential lung donor. Thorac Surg Clin 2022; 32:143-51. Venkateswaran
- Kotlo RM, Blosser S, Fulda GJ et al. Management of the potential organ donor in the ICU: Society of Critical Care Medicine/American College of Chest Physicians/Association of Organ Procurement Organizations consensus statement. Crit Care Med 2015;43(6):12911325,
- Hergenroeder GW, Ward NH, Yu X et al. Randomized trial to evaluate nutritional status and absorption of enteral feeding after brain death. Prog Transplant 2013;23(4):374-82.
- Carrott P, Cherry-Bukowiec JR, Jones CM et al. Nutrition therapy in the organ donor: theoretical benefits and barriers to implementation. Curr Nutr Rep 2016; 5:199–203.
- Torre-Cisneros J, Aquado JM, Caston JJ et al. Management of cytomegalovirus infection in solid organ transplant recipients: SET/GESITRA-SEIMC/ REIPI recommendations, Transplant Rev (Orlando) 2016;30(3):119-43,
- Anesi JA, Lautenbach E, Han J et al. Antibiotic utilization in deceased organ donors. Clin Infect Dis 2021;73(7):1284–7,
- Shemie SD, Ross H, Pagliarello J et al. Organ donor management in Canada: recommendations of the forum on Medical Management to Optimize Donor Organ Potential. CMAJ 2006;14;174(6):S13-S30.