UDK: 616.712-089.87-009.7-085.212
Kokareva A.
1University Clinic for Traumatology, Orthopedic Disease, Anesthesiology, Reanimation and Intensive Care Medicine, and Emergency Department, Faculty of Medicine, Ss. Cyril and Methodius University, Skopje, RN Macedonia
Abstract
Thoracotomy is associated with severe postoperative pain and a high incidence of chronic post-thoracotomy pain syndrome (CPTPS). Inadequate pain control adversely affects respiratory function, delays mobilization, increases postoperative complications, and contributes to long-term morbidity. Contemporary analgesic strategies emphasize a multimodal approach that integrates regional analgesic techniques, systemic pharmacotherapy, and adjuvant agents to optimize pain control while minimizing opioid consumption. This review provides a comprehensive overview of the pathophysiology of post-thoracotomy pain and critically analyzes current evidence supporting multimodal analgesic strategies in thoracic surgery.
Keywords: multimodal; pain management; post-thoracotomy pain syndrome; thoracotomy.
Introduction
Postoperative pain following thoracotomy remains one of the most challenging problems in perioperative medicine. Despite advances in surgical techniques, anesthetic management, and perioperative care, open thoracotomy is still associated with intense postoperative pain due to extensive tissue injury, rib retraction, intercostal nerve trauma, and pleural irritation. Poorly controlled pain results in impaired respiratory mechanics, reduced tidal volumes, ineffective coughing, and delayed mobilization, thereby increasing the incidence of pulmonary complications such as atelectasis, pneumonia, and respiratory failure (1-4). Beyond its immediate impact, severe acute pain is a well-recognized risk factor for the development of chronic post-thoracotomy pain syndrome, defined as pain persisting for more than two to three months after surgery. The reported incidence of CPTPS ranges from 25% to 60%, with neuropathic features present in a substantial proportion of patients. These long-term consequences have prompted a shift away from opioid-centric analgesia toward multimodal, opioid-sparing strategies that target multiple pain pathways simultaneously. Multimodal analgesia is now considered a cornerstone of modern thoracic anesthesia and enhanced recovery after surgery (ERAS) programs (1,5-8).
Pathophysiology of Post-Thoracotomy Pain
Post-thoracotomy pain is multifactorial and results from the interaction of peripheral and central mechanisms. Nociceptive pain arises from surgical trauma to the skin, subcutaneous tissue, muscles, ribs, and pleura, leading to activation of peripheral nociceptors and inflammatory mediator release. Visceral pain originates from manipulation of pulmonary and mediastinal structures (7). Neuropathic pain represents a critical component of post-thoracotomy pain and is frequently caused by direct injury, stretching, or compression of the intercostal nerves during rib retraction. Such nerve injury may result in ectopic discharges, altered ion channel expression, and long-lasting changes in neural signaling. Persistent nociceptive input can further induce central sensitization at spinal and supraspinal levels, characterized by amplified pain responses and reduced pain thresholds. These mechanisms explain the high incidence of chronic pain after thoracotomy and emphasize the need for early, mechanism-based analgesic interventions (8,9).
Concept of Multimodal Analgesia
Multimodal analgesia is defined as planned and simultaneous application of multiple analgesic techniques and pharmacological agents that act through different and complementary mechanisms of action. The primary objectives of this approach are to enhance overall analgesic efficacy through synergistic effects, reduce perioperative opioid consumption, minimize opioid-related adverse events, and promote early functional recovery (1,3).
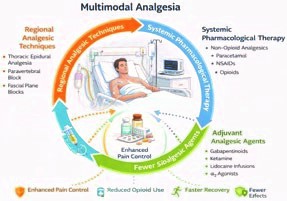
In the context of thoracic surgery, multimodal analgesia represents a cornerstone of modern perioperative pain management due to the intensity and complexity of post-thoracotomy pain. Effective multimodal strategies address both nociceptive and neuropathic pain components while limiting the physiological stress response to surgery. By targeting pain pathways at multiple levels, this approach improves respiratory mechanics, earlier mobilization, and reduced postoperative pulmonary complications (4). Implementation of multimodal analgesia in thoracic surgery spans all phases of the perioperative period. Preoperative measures include thorough patient education, risk stratification, and the use of pre-emptive analgesic interventions aimed at reducing central sensitization. Intraoperatively, emphasis is placed on regional anesthetic techniques and opioid-sparing anesthetic strategies to optimize pain control while preserving respiratory function. Postoperatively, management focuses on the scheduled administration of non-opioid analgesics, continuation of regional analgesia when applicable, and the judicious use of rescue opioids for breakthrough pain (1,8). This comprehensive, phase-specific approach is fully aligned with Enhanced Recovery After Surgery (ERAS) principles and has been consistently associated with improved clinical outcomes, including superior pain control, reduced opioid-related complications, shorter hospital stays, and enhanced patient satisfaction (Figure 1.) (1).
Figure 1. Multimodal analgesia
Regional Analgesic Techniques
Regional analgesic techniques play a pivotal role in multimodal pain management following thoracotomy by effectively interrupting afferent nociceptive transmission at the spinal and paraspinal levels. By directly targeting pain pathways, regional anesthesia provides superior analgesia, facilitates effective coughing and deep breathing, and supports early mobilization—key factors in optimizing postoperative recovery after thoracic surgery (3,6).
Thoracic Epidural Analgesia
Thoracic epidural analgesia has traditionally been regarded as the gold standard for postoperative pain control following open thoracotomy. By achieving a segmental blockade of thoracic spinal nerves, epidural analgesia provides excellent static and dynamic pain relief, particularly during respiratory efforts such as coughing and physiotherapy. This technique has been consistently associated with improved postoperative pulmonary function, enhanced patient comfort, and reduced incidence of pulmonary complications. Despite its well-established efficacy, the use of thoracic epidural analgesia may be limited by potential adverse effects related to sympathetic blockade, including hypotension, urinary retention, and pruritus. In addition, contraindications such as coagulopathy, systemic or local infection, and technical challenges may restrict its applicability in certain patient populations (3,6,10,11).
Paravertebral and Fascial Plane Blocks
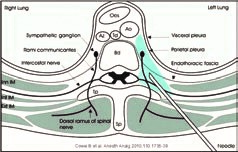
Thoracic paravertebral block offers effective unilateral segmental analgesia by anesthetizing spinal nerves as they emerge from the intervertebral foramina. Accumulating evidence indicates that paravertebral block provides analgesic efficacy comparable to thoracic epidural analgesia while demonstrating a more favorable side-effect profile, particularly with respect to hypotension and urinary retention (Figure 2). As a result, paravertebral block is increasingly regarded as a viable alternative to epidural analgesia in thoracic surgery (4,8). In recent years, ultrasound-guided fascial plane blocks, such as the erector spinae plane block, have gained significant attention due to their technical simplicity, reproducibility, and favorable safety profile. These techniques allow for effective analgesia with minimal risk of neuraxial complications and are particularly well suited for minimally invasive thoracic procedures and for patients in whom neuraxial anesthesia is contraindicated. When integrated into a comprehensive multimodal analgesic regimen, fascial plane blocks contribute substantially to opioid-sparing analgesia and improved postoperative recovery (4,12).
Figure 2. Paravertebral block
Systemic Pharmacological Therapy
Systemic pharmacological therapy constitutes a fundamental component of multimodal analgesia and serves as an essential adjunct to regional analgesic techniques. When used in combination, systemic agents enhance overall analgesic efficacy, address multiple pain pathways, and contribute significantly to opioid-sparing strategies in the postoperative period (13,14).
Non-opioid Analgesics
Paracetamol and non-steroidal anti-inflammatory drugs (NSAIDs) represent the cornerstone of systemic analgesia within multimodal pain management protocols. Through inhibition of cyclooxygenase enzymes and subsequent reduction in prostaglandin synthesis, these agents effectively decrease inflammation and peripheral nociceptor sensitization. When administered on a scheduled, rather than on-demand basis, paracetamol and NSAIDs have consistently demonstrated a significant reduction in postoperative opioid consumption and improved analgesic outcomes. Their favorable safety profile further supports their routine use, provided that patient-specific contraindications are carefully considered (3,8).
Opioids
Opioids remain potent and effective agents for the management of moderate to severe postoperative pain, particularly in the immediate post-thoracotomy period. However, within a multimodal analgesic framework, opioids should be used judiciously and primarily reserved for the treatment of breakthrough pain. Administration at the lowest effective dose is recommended to mitigate well-recognized adverse effects, including respiratory depression, postoperative nausea and vomiting, ileus, sedation, and the development of opioid-induced hyperalgesia. Limiting opioid exposure is especially important in thoracic surgery patients, in whom adequate respiratory function is critical for optimal recovery (3,8,13).
Adjuvant Agents
Adjuvant analgesic agents play a pivotal role in targeting neuropathic pain components and modulating central sensitization. Gabapentinoids reduce neuronal excitability through modulation of voltage-gated calcium channels and have been shown to decrease postoperative pain intensity and opioid requirements. Low-dose ketamine, acting as an N-methyl-D-aspartate (NMDA) receptor antagonist, effectively attenuates central sensitization and may reduce the risk of chronic post-thoracotomy pain. Intravenous lidocaine infusions provide additional analgesic, anti-inflammatory, and anti-hyperalgesic effects, contributing to improved pain control and faster functional recovery. Furthermore, α2-adrenergic agonists offer sedative, sympatholytic, and opioid-sparing benefits, thereby enhancing analgesic quality while preserving respiratory stability (4,9,14).
Prevention of Chronic Post-Thoracotomy Pain
Preventing the transition from acute to chronic pain is a primary objective of postoperative pain management. High-intensity acute pain, intercostal nerve injury, and inadequate early analgesia are key risk factors for CPTPS. Multimodal analgesic strategies incorporating effective regional anesthesia and early use of adjuvant agents have been shown to reduce both the incidence and severity of chronic pain.
Integration of multimodal analgesia into ERAS pathways further enhances recovery by promoting early mobilization, improved respiratory function, and reduced length of hospital stay. Emerging evidence suggests that such protocols may also improve long-term patient-reported outcomes and quality of life (1,14).
Conclusion
A multimodal approach is acknowledged as the most effective strategy for managing post-thoracotomy pain due to the intricate and multifaceted nature of pain linked to thoracic surgical procedures. Multimodal analgesia achieves superior pain control by integrating regional analgesic techniques such as: thoracic epidural analgesia, paravertebral blocks, or novel fascial plane blocks, with systemic pharmacological therapies, including non-opioid analgesics and judicious opioid use, compared to single modality strategies. This method diminishes overall opioid use and its related side effects, while enhancing respiratory mechanics, promoting earlier mobilization, and decreasing the occurrence of postoperative pulmonary complications. Moreover, efficient multimodal pain management has been linked to a diminished risk of persistent post-thoracotomy pain syndrome, thereby enhancing long-term functional results and quality of life. Due to the variety in patient features, surgical methods, and institutional resources, multimodal analgesic regimens must be customized to each patient’s clinical profile. Consequently, tailored multimodal pain management procedures ought to be regarded as fundamental and standard practice in modern thoracic anesthesia.
References:
- Mijatovic D, Knezevic J, Mijatovic M. Post-thoracotomy analgesia: current concepts and future directions. Saudi J Anaesth. 2021;15(3):328-336.
- Jovanovski-Srceva M, Kokareva A, Brzanov-Gavrilovska A. Factors affecting the occurrence of hypoxemia during one-lung ventilation. Pril (Makedon Akad Nauk Umet Odd Med Nauki). 2025;46(3):113–120. doi:10.2478/prilozi-2025-0028..
- Goto T. Perioperative pain management in thoracic surgery. J Thorac Dis. 2018;10(Suppl 4):S389-S401.
- Miyazaki T, Sakai T, Yamasaki N. Multimodal analgesia and prevention of chronic pain after thoracic surgery. J Thorac Dis. 2024;16(2):742-754.
- Humble SR, Dalton AJ, Li L. A systematic review of therapeutic interventions to reduce acute and chronic post-surgical pain after thoracotomy. Eur J Pain. 2015;19(4):451-464.
- Gerner P. Post-thoracotomy pain management. Anesthesiol Clin. 2008;26(2):355-367.
- Wildgaard K, Ringsted TK, Kehlet H. Chronic post-thoracotomy pain: a critical review of pathogenic mechanisms and prevention. Eur J Cardiothorac Surg. 2009;36(1):170-180.
- Peng K, Liu HY, Wu SR, et al. Multimodal analgesia strategies in thoracic surgery: a systematic review. J Healthc Eng. 2022;2022:1-10.
- Mori S, Hasegawa T, Endo T. Risk factors and prevention of chronic post-thoracotomy pain. Nagoya J Med Sci. 2022;84(3):401-410.
- Mehta Y, Arora D, Mehta C. Regional analgesia and multimodal pain management in thoracic surgery. BJA Educ. 2023;23(3):79-86.
- Taleska G, Trajkovska T, Kokareva A, et al. Preemptive epidural analgesia with bupivacaine and sufentanyl and the effects of epidurally added epinephrine for thoracic surgery. Maced J Med Sci. 2010;3(1):XXX–XXX. doi:10.3889/MJMS.1957-5773.2010.0092.
- Cowie B, McGlade D, Ivanusic J, Barrington MJ. Ultrasound-guided thoracic paravertebral blockade: a cadaveric study. Anesth Analg. 2010;110:1736–1739.
- Baser O, Erdem AF, Yilmaz C. Opioid-sparing multimodal analgesia in thoracic surgery: outcomes and clinical implications. BMC Anesthesiol. 2025;25:112.
- Kokareva A, Brzanov-Gavrilovska A, Kondov G, Jovanova S, Buntashevska B, Jovanovski-Srceva M. One-lung ventilation and its role in postoperative pulmonary morbidity following lung surgery. MJA,2024;8(4):35-45.