UDK: 616.714.1-089.85-009.7-089.5
Naumovski F.
1University Clinic for Traumatology, Orthopedic Diseases, Anesthesia, Reanimation, Intensive
Care and Emergency Centre in Skopje; Faculty of Medicine, SS Cyril and Methodius University,
Skopje
Abstract
Almost 90% of patients who undergo craniotomy experience postoperative pain that is poorly controlled in the early postoperative period. As many as half of them are considered to have moderate to severe headaches caused by the surgical procedure. The use of opiates in the intraoperative and postoperative period is the most common method for pain management. Scalp block is a regional anesthetic technique in which local anesthetic is infused near the nerves that innervate the scalp, thus providing quality intraoperative and postoperative analgesia. When applying the scalp block, 2 ml of local anesthetic is infiltrated at 5 sites on the side where the craniotomy will be performed. The scalp block blocks n.supraorbitalis and n.supratrochlearis; n.auriculotemporalis; n.occipitalis major and n.occipitalis minor. When applying the scalp block, long-acting local anesthetics such as bupivacaine or ropivacaine are used, while a combination of lidocaine and bupivacaine is also possible. Systemic application of corticosteroids prolongs the analgesic effect of the block for up to 48 hours, and according to some authors, up to 72 hours. The scalp block drastically reduces the need for opiates in the intraoperative period and provides complete hemodynamic stability (1). According to the meta-analysis by D. Taylor and colleagues, scalp blocks reduce opioid consumption in the first 24 and 48 hours postoperatively, and also significantly reduces pain by 2/10 in the first 24 hours after the surgical procedure, with the possibility of extending the analgesic effect for up to 48 hours (2)(3). Additionally, scalp block significantly or completely attenuates the hemodynamic effects of craniotomy, providing hemodynamic stability immediately after its application (2)(3). According to the ESAIC recommendations for the management of craniotomy-related pain issued in 2023, analgesia for craniotomy should consist of scalp block combined with intraoperative administration of paracetamol, with or without continuous infusion of dexmedetomidine, while opiates are considered only as salvage therapy when all other modalities have failed (4).
Keywords: Craniotomy; Scalp Block; Regional Anesthesia.
Introduction
Craniotomy as a surgical procedure can be a cause for postoperative pain in approximately 8090% of patients (1,2). Unfortunately, postoperative pain after craniotomy has been neglected and treated insufficiently worldwide due to clinical underestimation of the pain severity, as well as the risk of sedation when using opioids as a rescue technic in pain control. It is well known that early postoperative pain control could be key to preventing chronic pain development and central sensitization. Therefore, perioperative pain management in either elective or urgent craniotomies is more than important, while at the same time, the patients are guided safely through the therapeutic process. The pain resulting from neurosurgical interventions is clearly somatic by origin, resulting from the skin, muscle and bone injuries that have occurred due to the surgical intervention. However, the pain is not visceral at all because it is well known that the pain is not caused by damaging the brain itself. It was reported that as many as half of previously mentioned 90% of patients are considered to have moderate to severe headache caused by the surgical procedure (1). Since, the percentage of patients experiencing severe postoperative pain is serious and clinically relevant, the discussion in this article will focus on the usage of regional anesthesia as a technique for pain control, providing precise data about the usage of scalp block in daily practice with careful review of latest recommendations with relevant clinical implications.
Craniotomy-related Pain
As previously said, the pain related to craniotomy is purely somatic, originating from the damage of the skin of the scalp, muscles, periost and the bone itself, as well as the dissection of the meninges. However, the pain is not visceral because the brain itself does not possess pain receptors. Regardless the type of craniotomy, the pain is usually concentrated around the incision site and not involving parts of the head which were not affected by the surgical incision. The pain stimuli are created usually because of incision, dissection and retraction of tissues involving dermatomes in the innervational area of the trigeminal nerve, as well as branches from the cervical plexus. Temporal craniotomy, long lasting surgeries, subtle nerve damage and implantation of osteosynthetic material were considered risk factors that could worsen the pain. Given that the pain itself is a negative postoperative experience that was left undertreated in daily practice, it is worth mentioning that the pain arising from craniotomy is related to surge of catecholamines due to activation of the stress responses related to pain. Actually, pain-related stress response and following catecholamine release are strongly unwanted, since the hemodynamic disturbances in the perioperative period could end with undesirable complications and hemorrhage leading to an unfavorable outcome. Evidently, the importance of strict perioperative pain management in patients undergoing craniotomy does not consist merely of pain control, rather securing a hemodynamic stability.
SCALP Block: How to Perform It?
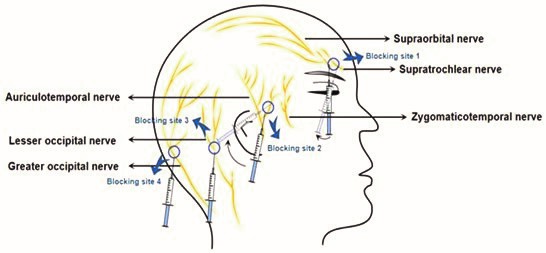
The head possesses dual sensitive innervation, arising from the trigeminal nerve and cervical plexus. In fact, to provide full analgesia of the skin, muscles and the periost, the anesthetist must know the innervational area of each one of the nerves that provide sensitive innervation of the head. Therefore, performing a scalp block implies infiltrating local anesthetic in a wellknown landmark guided point around the following nerves: supraorbital and supratrochlear nerve, zygomaticotemporal and auriculotemporal nerve, as well as minor and major occipital nerve, together with magnus occipital nerve. Providing successful intraoperative and postoperative analgesia is possible with infiltration of long-lasting local anesthetic on a few sites guided by well-known anatomical landmarks without the need of ultrasound. Application of 2ml of plain bupivacaine or ropivacaine at each site provides immediate analgesia. The first application
Macedonian Journal of Anaesthesia
site is located above the orbit in the middle where the exit of the supraorbital nerve is located. Anatomically, it is recognized by the incisura on the upper edge of the orbit, which should be palpated. The 2ml of local anesthetic should be infiltrated immediately above, thus blocking the supraorbital nerve. Using the same puncture, but only by changing the direction of the needle medially towards the nose, another 2ml of local anesthetic should be infiltrated, thus blocking the supratrochlear nerve. With one puncture and two different infiltrations, analgesia is provided for the frontal and parietal regions. Then, just before the tragus, the pulse of the auriculotemporal artery is palpated preauricularly and carefully, without injuring it, while another 2 ml is applied around the artery, thus blocking the auriculotemporal nerve. Then, through the same puncture, the direction is changed to the medial area, where 2 ml is applied, thus blocking the zygomaticotemporal nerve. By blocking these two nerves, analgesia of the temporoparietal part of the scalp is provided. Next, at a distance of 2-3 cm lateral to the external occipital protuberance, another 2 ml of local anesthetic is infiltrated, to block the greater occipital nerve. The lesser occipital nerve is blocked by infiltrating 1-2 ml of local anesthetic medial to the mastoid and at the posterior border of the sternocleidomastoid, which blocks the lesser occipital nerve. The lesser and greater occipital nerves provide sensory innervation of the occipital part of the scalp. The same application sites apply to the application of this block in pediatric neuroanesthesia, so it is necessary to pay great attention to the dose of local anesthetic and adjust it according to the patients’ weight and the reduced level of acid alpha glycoprotein.
Figure1 Injection sites according to the anatomical dispersion of nerves
The Science Behind Scalp Block
In the past, craniotomy-associated pain in the intraoperative and early postoperative period was most often managed with the use of opioids. The modern approach involves reducing opioids to the minimum possible level, taking into account their adverse effects such as drowsiness, impaired consciousness, respiratory depression, nausea and vomiting, as well as the presence of miosis, which can interfere with early and timely recognition of signs of increased intracranial pressure or the occurrence of brain edema. Hence, the application of regional anesthetic techniques in everyday neurosurgical technique overshadows opioids as the first line of choice in the treatment of craniotomy-associated pain. Hence, scalp block is defined as a sovereign, modern, safe, easy to perform, and simple regional anesthetic technique for providing adequate intraoperative and postoperative analgesia. If a long-acting anesthetic is used for the block in combination with dexamethasone or dexmedetomidine, they will prolong the effect of the block (3). The superiority of scalp block in managing craniotomy-related pain is also confirmed by the results of a meta-analysis, according to which patients who received scalp block had significantly lower opioid requirements in the first 24 hours after surgery and less pain compared to patients who did not receive scalp block (4). Also, according to the authors of the same meta-analysis, scalp block provided significantly better hemodynamic stability during the intraoperative period, from the moment of the first incision until the end of the operation, with significantly lower heart rate and mean arterial pressure values a nd without strokes. These effects were observed in the intraoperative period in patients who received scalp block therapy (4). The positive effects of scalp block are confirmed by another meta-analysis based on data from 1500 patients, which came to a clear conclusion regarding the usefulness and the impact of scalp block on the occurrence of pain and on the stress response. Namely, patients who received scalp block had satisfactory analgesia up to 48 hours after surgery, but also a delayed time to require rescue analgesia after the end of the surgery, compared to the control group where scalp block was not applied (5). This meta-analysis comes to much more precise conclusions about the impact of scalp block on the occurrence of stress response during craniotomy. The reviewed studies determined intraoperatively and postoperatively much lower levels of cortisol and angiotensin in patients where scalp block was applied compared with the control group. These findings explicitly highlight the effect of regional anesthesia on systemic hemodynamics (5). Numerous authors point to the analgesic power of scalp block in the early postoperative period, but limit it to 24 hours, while Duda and colleagues in their meta-analysis conclude that scalp block provides analgesia for up to 72 hours and that it significantly reduces opioid consumption 24 and 48 hours after surgery (6). In that context, in regards to postoperative pain, another meta-analysis found superiority of scalp block application only in the early and the middle period of the first 24 hours, but not in the late first 24 hours, with patients who received the block having significantly lower pain scores only in the early and middle hours after the intervention compared to the control group, which was not the case in the late hours of the first postoperative day (7). The difference between one and the other meta-analysis may be due to the heterogeneity of the results incorporated in the second meta-analysis, according to which the use of scalp block was associated with lower pain scores only in the first half of the first 24 hours after surgery. Another meta-analysis also confirms the analgesic effect of scalp block in the first 24 hours, noting that it is associated with lower VAS in the first 24 hours, lower overall analgesics usage, and a significantly longer time for administration of rescue analgesia, compared to the control group where scalp block was not applied (8). In order to provide sufficient and successful analgesia when applying scalp block long lasting anesthetics as bupivacaine, levobupivacaine and ropivacaine are recommended. Howerver, some authors state that ropivacaine is superior when compared with others (9). Compared with local anesthetic infiltration, scalp block provides better analgesia according to one randomized trial (9). Previously, other authors have also published results according to which the application of scalp block is superior to the application of local anesthetic only to the incision site. It is likely that the selective blockade of the trigeminal and occipital nerve fibers provides deeper and longer-lasting analgesia and also appears to have anti-inflammatory properties. It is also probable that the selective blockade has a suppressive effect on the stress response, which likely plays an important role in the development of postoperative pain following craniotomy. It is also important to note that when it comes to pain control the application of the block before surgical incision is superior to its application postoperatively. Additionally, but no less important, the application of the scalp block significantly reduces the intraoperative use of opioids regardless of whether it is fentanyl or remifentanil, which is a significant fact that
Macedonian Journal of Anaesthesia
completely changes the concept of the use of fentanyl anesthesia in neurosurgery. Moreover, patients who were anesthetized with a scalp block not only received a smaller amount of opiates compared to patients who did not receive a scalp block, but also had identical or even better hemodynamic stability compared to the opioid group (9). Hence, it seems that the introduction of this regional anesthetic technique into everyday neuroanesthesia can not only reduce everyday opiate usage, but can also contribute to faster and easier awakening from anesthesia without the presence of opioid side effects.
Guidelines and Recommendations
According to the PROSPECT study recommendations that are the newest official document published by ESAIC, the management of postoperative pain after elective craniotomy may be complex and should be multimodal. They specify each of the available medications and their effect when dealing with postoperative pain after craniotomy. According to the PROSPECT study, application of the scalp block is the foundation in providing quality perioperative analgesia and preventing central sensitization of the pain. The study strongly recommends preincisional application of the scalp block in order to provide sufficient analgesia. According to PROSPECT study team, the application of scalp block offers long lasting analgesia from 2-48 hours after surgery with significantly reduced postoperative opioid consumption (10). Despite the clearly stated opioid sparing effects of scalp block, according to the study team, its application is related to less frequent postoperative nausea and vomiting. Finally, according to the PROSPECT recommendations, scalp block should be routinely used in patients that undergo elective craniotomy in combination of paracetamol, NSAID and dexamethasone and/or dexmedetomidine, while opioids should be used only as a rescue analgesia (10).
Conclusion
The scalp block is a simple to perform, easy, safe and sovereign regional anesthetic technique that provides adequate intraoperative and postoperative analgesia in elective and emergency craniotomies. The use of this technique is associated with significantly reduced perioperative opioid use and reduced incidence of opioid-associated complications. The scalp block is part of a modern multimodal approach to the treatment of craniotomy-associated pain and is recommended by ESAIC as the technique of choice and first line of treatment before opioid use.
References:
- Moha rari RS, Emami P, Neishaboury M, Sharifnia SH, Kianpour P, Hatam M, et al. Scalp nerve block for enhanced pain control and analgesic optimization in elective craniotomy: a randomized controlled trial with analgesia nociception index monitoring. World Neurosurg. 2024;189:e55–e60. doi:10.1016/j.wneu.2024.05.144.
- Luo M, Zhao X, Deng M, Hu Y, Yang X, Mei Z, et al. Scalp nerve block, local anesthetic infiltration, and postoperative pain after craniotomy: a systematic review and network meta-analysis of randomized trials. J Neurosurg Anesthesiol. 2023;35(4):361–374. doi:10.1097/ANA.0000000000000868.
- Chen Y, Ni J, Li X, Zhou J, Chen G. Scalp block for postoperative pain after craniotomy: A meta-analysis of randomized control trials. Front Surg. 2022 Sep 26;9:1018511. doi: 10.3389/fsurg.2022.1018511. PMID: 36225222; PMCID: PMC9550001.
- Bombardieri AM, Pochebyt M, Burbridge MA. Update on scalp nerve block for craniotomy. Curr Opin Anaesthesiol. 2025 Oct 1;38(5):598-604. doi: 10.1097/ACO.0000000000001546. Epub 2025 Jun 27. PMID: 40548352.
- Fu PH, Teng IC, Liu WC, Chen IW, Ho CN, Hsing CH, Sun CK, Hung KC. Association of scalp block with intraoperative hemodynamic profiles and postoperative pain outcomes at 24-48 hours following craniotomy: An updated systematic review and meta-analysis of randomized controlled studies. Pain Pract. 2023 Feb;23(2):136-144. doi: 10.1111/ papr.13167. Epub 2022 Oct 6. PMID: 36176201.
- Wei X, Liu Z, Liu C, Li S, An J, Wang Z. The effect of scalp nerve block on postoperative analgesia and stress response in patients undergoing craniotomy: a meta-analysis. Altern Ther Health Med. 2024;30(10):179–187.
- Duda T, Lannon M, Gandhi P, Martyniuk A, Farrokhyar F, Sharma S. Systematic review and meta-analysis of randomized controlled trials for scalp block in craniotomy. Neurosurgery. 2023;93(1):4–23. doi:10.1227/neu.0000000000002381.
- Wardhana A, Sudadi S. Scalp block for analgesia after craniotomy: A meta-analysis. Indian J Anaesth. 2019 Nov;63(11):886-894. doi: 10.4103/ija.IJA_315_19. Epub 2019 Nov 8. PMID: 31772396; PMCID: PMC6868657.
- N S, V AP, Kateel R, Balakrishnan A, Nayak R, Menon GR, M S, Bhat R. Posterior scalp block with bupivacaine and dexmedetomidine for pain management in posterior fossa surgeries: a prospective, double blind randomized controlled trial. Pain Manag. 2025 Mar;15(3):131-140. doi: 10.1080/17581869.2025.2470607. Epub 2025 Mar 1. PMID: 40022547; PMCID: PMC11881862.
- Mestdagh FP, Lavand’homme PM, Pirard G, Joshi GP, Sauter AR, Van de Velde M; PROSPECT Working Group of the European Society of Regional Anaesthesia and Pain Therapy (ESRA). Pain management after elective craniotomy: A systematic review with procedure-specific postoperative pain management (PROSPECT) recommendations. Eur J Anaesthesiol. 2023 Oct 1;40(10):747-757. doi: 10.1097/EJA.0000000000001877. Epub 2023 Jul 6. PMID: 37417808.